Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications

Selective methodologies for the synthesis of biologically active piperidinic compounds - Cossy - 2005 - The Chemical Record - Wiley Online Library
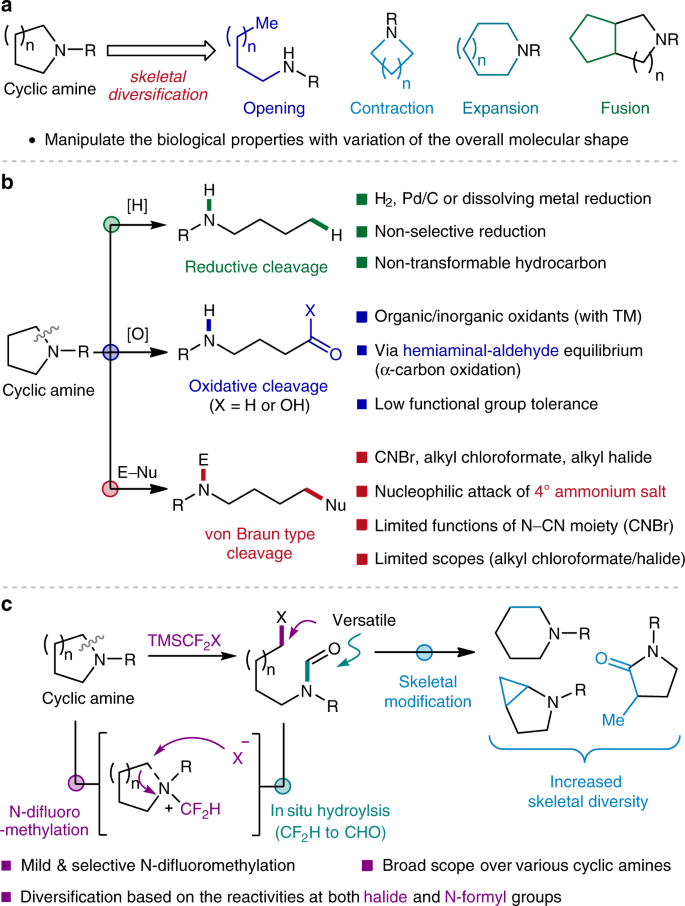
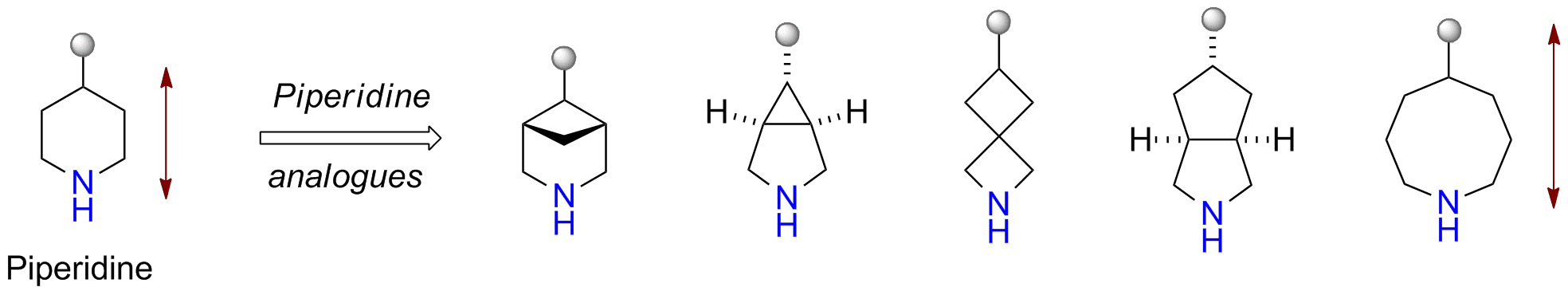
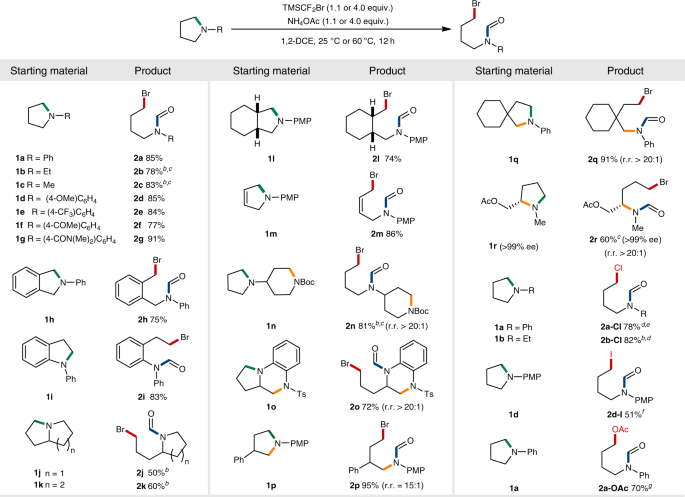
Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications
![EP1002794A1 - 4-[(Aryl)(aryloxy)methyl]piperidine derivatives and their use as serotonin and/or noradrenaline reuptake inhibitors - Google Patents EP1002794A1 - 4-[(Aryl)(aryloxy)methyl]piperidine derivatives and their use as serotonin and/or noradrenaline reuptake inhibitors - Google Patents](https://patentimages.storage.googleapis.com/27/d2/a6/f0da448169f65c/00020001.png)
EP1002794A1 - 4-[(Aryl)(aryloxy)methyl]piperidine derivatives and their use as serotonin and/or noradrenaline reuptake inhibitors - Google Patents

Benzyne‐Induced Ring Opening Reactions of DABCO: Synthesis of 1,4‐Disubstituted Piperazines and Piperidines - Seo - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

Recent progress toward the asymmetric synthesis of carbon-substituted piperazine pharmacophores and oxidative related heterocycles - RSC Medicinal Chemistry (RSC Publishing)
Welcome to Chem Zipper.com......: Pyridine is almost 1 million times less basic than piperidine? Why?

Fentanyl-related compounds and derivatives: current status and future prospects for pharmaceutical applications. - Abstract - Europe PMC
Applied Sciences | Free Full-Text | Hydrogen Storage: Thermodynamic Analysis of Alkyl-Quinolines and Alkyl-Pyridines as Potential Liquid Organic Hydrogen Carriers (LOHC) | HTML
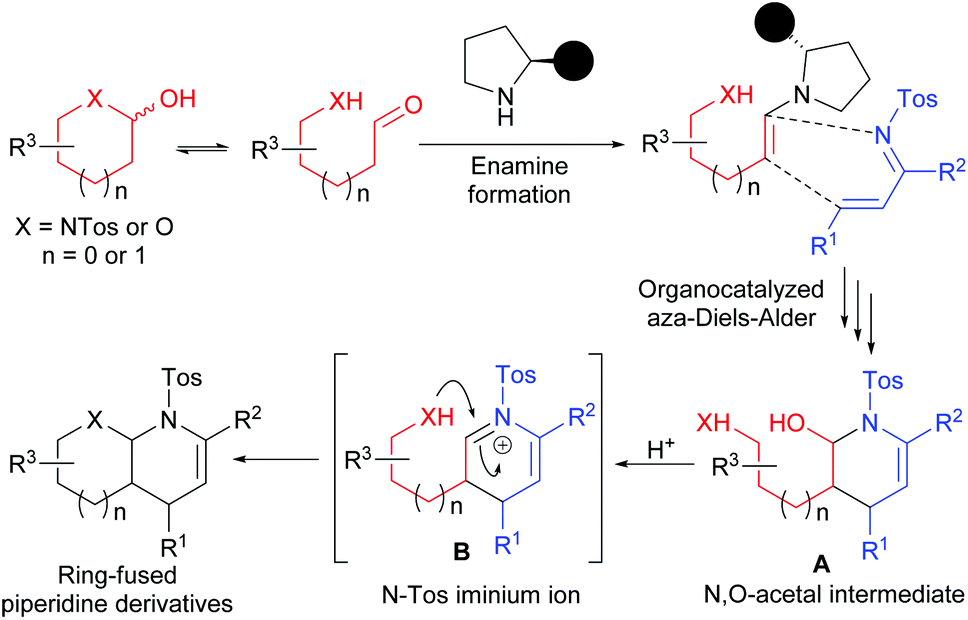
One-pot, highly efficient, asymmetric synthesis of ring-fused piperidine derivatives bearing N , O - or N , N -acetal moieties - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB02571K
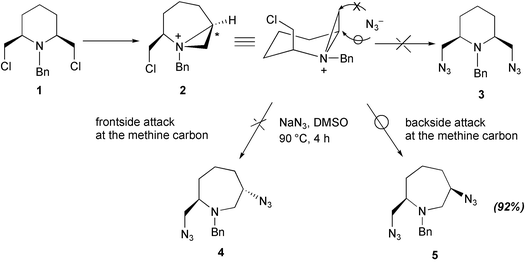


/DB19273E6550AD06802585F9007F9729/$file/FP36451_structure.png)